
We established that several host innate immune signaling proteins are required for the observed macrophage activation, including the Card9 and MyD88 adaptor proteins, as well as the Dectin-1 and TLR2 pattern recognition receptors. These cell surface changes influence the host-pathogen interaction, resulting in increased macrophage activation to microbial challenge in vitro. We observed that the Mar1 protein differentially localizes to cellular membranes in a condition dependent manner, and we have further shown that the mar1Δ mutant displays defects in intracellular trafficking, resulting in a mislocalization of the β-glucan synthase catalytic subunit, Fks1. However, HPLC analysis of whole cell walls and RT-PCR analysis of cell wall synthase genes demonstrated that this increased chitin exposure is likely due to decreased levels of glucans and mannans in the outer cell wall layers. 
Furthermore, the mar1Δ mutant displays increased staining for exposed cell wall chitin and chitosan when the cells are grown in host-like tissue culture conditions. Through phenotypic studies of a loss-of-function strain, we have demonstrated that the mar1Δ mutant has an aberrant cell surface and a defect in polysaccharide capsule attachment, resulting in attenuated virulence. In a genetic screen for mutants with changes in their cell wall, we identified a novel protein, Mar1, that controls cell wall organization and immune evasion. 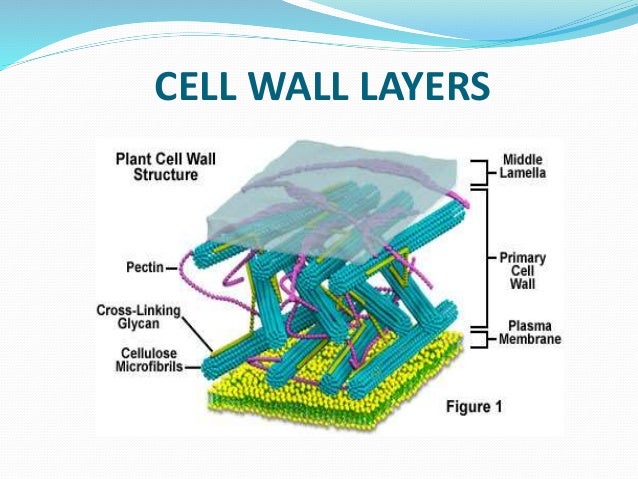
This cell wall remodeling is essential for host immune avoidance by this pathogen. The human fungal pathogen, Cryptococcus neoformans, dramatically alters its cell wall, both in size and composition, upon entering the host.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
